ExFlyBoy5
Give me a museum and I'll fill it. (Picasso) Give me a forum ...
Long story short...we live in a home that was built 15 years ago and is on a well. Over the last year we (and most of our other neighbors) have had issues with fittings (mostly brass) corroding and causing leaks. Most of the plumbers immediately declare "HARD WATER!" as a culprit but 2 of the neighbors have had water softeners installed from day one and still have the problem. As an aside, we have a 20 gallon pressure tank and digital pressure controller, so the system is always under pressure. We also use a hot water circulator pump about 18 hours out of the day.
Some research and basic chemistry understanding (from dealing with a swimming pool) led me in the direction of a low pH. Testing at the well head and interior faucets shows a pH of about 6.5 with a Taylor test kit as well as a pH meter. So, all signs pointed to getting an acid reducer to stop/slow down the process. Also, all the affected fittings also had the tell tale blue staining, especially around the cooper fittings.
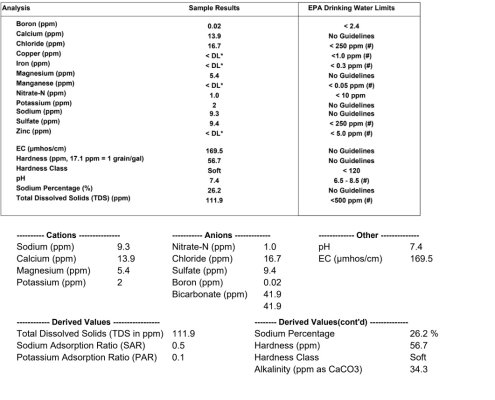
So, I took a water sample (actually two...drawn 48 hours apart) to our local university for testing (attached). All the tests show pretty normal numbers and a very normal pH. The only thing I can figure is that the low pH may have increased with handling and transport. No other numbers seem to show anything that would cause this type of corrosion.
So, for those of you with a well...or a good understanding of chemistry (mine stopped at the 10th grade level) what are your thoughts? I don't want to drop $1,000+ on an acid reducer that won't do any good.
Some research and basic chemistry understanding (from dealing with a swimming pool) led me in the direction of a low pH. Testing at the well head and interior faucets shows a pH of about 6.5 with a Taylor test kit as well as a pH meter. So, all signs pointed to getting an acid reducer to stop/slow down the process. Also, all the affected fittings also had the tell tale blue staining, especially around the cooper fittings.
So, I took a water sample (actually two...drawn 48 hours apart) to our local university for testing (attached). All the tests show pretty normal numbers and a very normal pH. The only thing I can figure is that the low pH may have increased with handling and transport. No other numbers seem to show anything that would cause this type of corrosion.
So, for those of you with a well...or a good understanding of chemistry (mine stopped at the 10th grade level) what are your thoughts? I don't want to drop $1,000+ on an acid reducer that won't do any good.
Attachments
Last edited: