Smoke Alarm Battery Utilization
A minor peeve: Why don't designers of battery-operated consumer devices make their stuff to use batteries efficiently?
One of our smoke alarms started the "low battery" beeping last night (it's always at night, right?). So, I put new AA alkaline batteries in all the detectors, and the three in the "offending" detector showed 1.25v, all the other detectors showed 1.3V
An AA alkaline battery showing 1.25V typically has used up only about 22% of its total capacity (.5 Ah of 2.3Ah, per graph below). Even if we assume a 1V cutoff voltage for the device, a cell showing 1.25 V has 75% of its usable capacity remaining (.5 Ah of 2.0 Ah). Why is the "low battery" chirp going off?
This device takes three batteries, wired in series. If it really can't operate below 3.75 v (1.25v x 3), then why not build it to take 4 batteries so they can all discharge down to 1.15V and we can get a lot more of the available capacity (twice as much) from all of them? Likewise, if the constraint is the need for sufficient onboard capacity to run the buzzer and RF transmitter (these are interlinked wireless alarms) when smoke is detected, having 4 batteries onboard and letting them discharge to a lower level would achieve the same result with less hassle for the user. As a bonus, with a 1.15V "chirp" voltage, the new low-self-discharge rechargeable NiMH batteries could be used (see the blue line below), they are a lot more economical and more "green" than alkaline batteries.
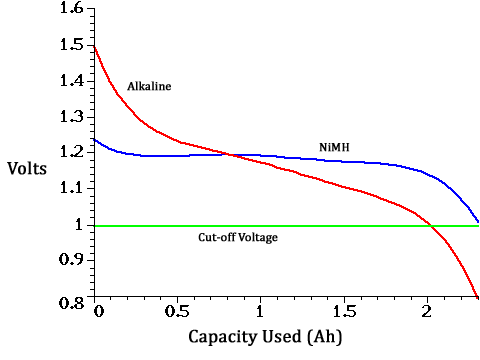
I suspect the reason the detectors are designed this way is that it is a conservative approach for a "must work" application. Change the three batteries every year and there's sure to be enough "oomph" in the batteries to get the job done, and that's the important thing. It doesn't matter if the users are throwing away batteries every year with 75% of their useful life remaining.
For now I'll just put these slightly-used batteries from the detectors into flashlights and other things that can use them.
.
.

